| April/May 2012 |
|
| January 2012 |
 |
|
Special Focus Issue 'Seed Biology' Plant Cell Physiology 53 (1) January 2012
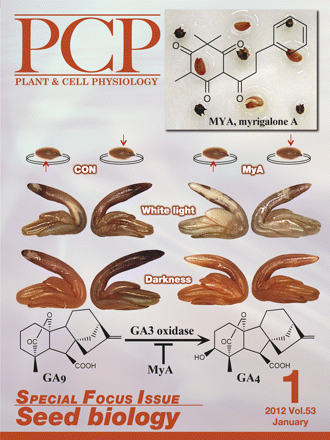
On the cover: Seeds are efficient plant dispersal units and to this end have evolved a number of unique survival strategies, including the ability to remain dormant without losing viability. The complex mechanisms of dormancy are probably the consequence of natural selections on mutations, or 'trials and errors', which seed-bearing plants have undergone during the course of evolution. The mechanisms controlling dormancy and seedling emergence via germination are regulated through changes in hormonal, metabolic, and gene expression patterns. This Special Focus Issue on Seed Biology introduces recent progress in these fields, and provides perspectives about future seed science research including its potential applications. One of the many topics in focus is seeds of parasitic weeds, which have adopted tactics to germinate on the host species when attracted by its exuded substance(s). In contrast, seeds or fruits can produce inhibitors to prevent seed germination in other species. The cover image reveals the effects of an allelochemical and putative natural herbicide, Myrigalone A (MyA), on seed germination. Upper right panel shows suppressed germination of garden cress (Lepidium sativum) seeds (light brown) by MyA leaked from sweet gale (Myrica gale) fruits (dark brown). Middle panel and scheme show nitroblue tetrazolium staining of superoxide radicals in cress embryos and active GA biosynthesis, both of which are affected by MyA (see Oracz et al.; page 81–95 for details). Cover design by Gerhard Leubner and Krystyna Oracz. |
 |
 |
 |
|
 |
|
| November 2011 |
Recent review on the regulation of seed germination by ethylene, jasmonates, and other hormones:
Beyond gibberellins and abscisic acid: how ethylene and jasmonates control seed germination
Linkies, Leubner-Metzger
Plant Cell Reports 31: 253-270 (2012)
|
 |
|
| April 2010 |
Recent review on the ecolution of plant seeds:
Tansley Review " The evolution of seeds"
Linkies, Graeber, Knight, Leubner-Metzger
New Phytologist 186: 817-831 (2010)
|
 |
|
| Fall 2009 |

Recent Press and Web articles:
ISSS Newsletter 16 (October 2009) including an article about "The Europaen Virtual Seed Network Project"
Hyperlink: vSEED - ERA-NET Plant Genomics "virtual SEED" - www.vseed.eu
Hyperlink: "vSEED" - BIOPRO - Das Biotech/Life Sciences Portal Baden-Württemberg
Hyperlink: "Modellorganismen" - BIOPRO - Das Biotech/Life Sciences Portal Baden-Württemberg
Hyperlink: "Samen im Tiefschlaf " - BIOPRO - Das Biotech/Life Sciences Portal Baden-Württemberg
|
 |
|
| November 2006 |
Recent reviews on the hormonal signaling network and the hormonal regulation of seed dormancy and germination:
Finch-Savage WE, Leubner-Metzger G (2006)
Seed dormancy and the control of germination.
Tansley review: New Phytologist 171: 501-523 (2006)
Seed dormancy is an innate seed property that defines the environmental conditions in which the seed is able to germinate. It is determined by genetics with a substantial environmental influence which is mediated, at least in part, by the plant hormones abscisic acid and gibberellins. In this review we present an integrated view of the evolution, molecular genetics, physiology, biochemistry, ecology and modelling of seed dormancy mechanisms and their control of germination. We argue that adaptation has taken place on a theme rather than via fundamentally different paths and identify similarities underlying the extensive diversity in dormancy response to the environment that controls germination.
Kucera B, Cohn MA, Leubner-Metzger G (2005)
Plant hormone interactions during seed dormancy release and germination.
Invited review: Seed Science Research 15: 281-307 (2005)
This review focuses on the interactions between abscisic acid (ABA), gibberellins (GA), ethylene, brassinosteroids (BR), auxin and cytokinins in regulating the interconnected molecular processes that control dormancy release and germination. ABA is a positive regulator of dormancy induction and most likely also maintenance, while it is a negative regulator of germination. GA releases dormancy, promotes germination and counteracts ABA effects. Ethylene and BR promote seed germination and also counteract ABA effects. We present an integrated view of the molecular genetics, physiology and biochemistry used to unravel how hormones control seed dormancy release and germination.
Table of plant hormone mutants in seed biology:
Comprehensive table of mutants (Arabidopsis and other species) affected in seed germination and/or dormancy due to mutations related to gibberellins, absicisc acid, ethylene or brassionsteroids. From Kucera et al. (2005).
Figure about plant hormone interactions in seed biology:
Schematic representation of the interactions between the gibberellin (GA), absisic acid (ABA) and ethylene signaling pathways in the regulation of seed dormancy and germination of Arabidopsis and other species. From Kucera et al. (2005).
|
 |
|
| May 2005 |
Redesign of the website "The Seed Biology Place" - now located at http://www.seedbiology.de |
 |
|
| September 2002 |
Year-2002 reviews on the hormonal signaling network and the hormonal regulation of seed dormancy and germination:
Comment: Plant hormones are of utmost importance in the regulation of the genetical, physiological and biochemical properties of seeds. Only little is known about the interconnected molecular key processes regulating seed dormancy and germination in response to plant hormones and environmental cues. The dry dormant seed is well equipped to survive extended periods of unfavorable conditions. Seed dormancy can be 'coat-imposed' and/or determined by the embryo itself and is a temporary failure or block of a viable seed to complete germination under physical conditions that normally favor the process (Koornneef et al. 2002). There is considerable genetic variation of seed dormancy within species, accessions of wild type and varieties of cultivated plants. The substantial influence of environmental effects on the expression of germination characteristics and the involvement of many genes make dormancy a typical quantitative trait, which is subject to QTL (quantitative trait loci) analysis (Koornneef et al. 2002). In many plant species with coat-imposed dormancy the seed envelope imposes a physical constraint to radicle protrusion, which has to be overcome by the growth potential of the embryo. In cases of endosperm-limited germination, weakening of the micropylar endosperm surrounding the radicle tip seems to be required for radicle protrusion and is likely to involve cell-wall hydrolysis by the action of hydrolytic enzymes. Major recent contributions to the issue of the hormonal regulation of endosperm-limited germination were achieved by using tomato and Nicotiana species as suitable model systems for endospermic seeds (e.g. Grappin et al., 2000; Koornneef et al. 2002; Leubner-Metzger 2000; Toorop et al,2000; Wu et al., 2000). However, recent progress in the field of seed biology was especially achieved by molecular genetic studies with Arabidopsis thaliana hormone mutants. Several recent reviews focus on the interactions between abscisic acid (ABA), gibberellins (GA), and brassinosteroids (BR) in regulating the interconnected molecular key processes that determine dormancy and germination. These reviews are listed here and the reading of these year-2002 highlights is highly recommended.
(1) Of special interest for seed biologists:
• Koornneef, M., Bentsink, L. and Hilhorst, H. (2002) Seed dormancy and germination.
Current Opinion in Plant Biology 5, 33-36 (hyperlink to abstract).
(2) Gibberellins (GA):
• Peng, J. and Harberd, N.P. (2002) The role of GA-mediated signalling in the control of seed germination.
Current Opinion in Plant Biology 5, 376-381 (hyperlink to abstract).
• Olszewski, N., Sun, T.P. and Gubler, F. (2002) Gibberellin signaling:
Biosynthesis, catabolism, and response pathways.
The Plant Cell 14, S61-S80 (no abstract, hyperlink to supplement issue).
(3) Brassinosteroids (BR):
• Bishop, G.J. and Koncz, C. (2002) Brassinosteroids and plant steroid hormone signaling.
The Plant Cell 14, S97-S110 (no abstract, hyperlink to supplemenr issue).
(4) Abscisic acid (ABA):
• Finkelstein, R.R., Gampala, S.S.L. and Rock, C.D. (2002) Abscisic acid signaling in seeds and seedlings.
The Plant Cell 14, S15-S45 (no abstract, hyperlink to supplement issue).
|
 |
|
| October 2001 |
 |
 |
The "Botanikertagung" is the conference of the "Deutsche Botanische Gesellschaft" (German Botanical Society) and took place in Freiburg i. Br. (Germany) in September 2002. The website of the "Botanikertagung Freiburg i. Br. 2002" is not any more availabe. However, the website section about the famous glas paintings of Friburg's cathedral are now available as part of this website. Klick on the hyperlink "Freiburg" in the navigation menu.
|
|
 |
|
| July 2001 |
Camille M. Steber and Peter McCourt
A role for brassinosteroids in germination in Arabidopsis
Plant Physiology 125: 763-769 (2001)
and
Leubner-Metzger G
Brassinosteroids and gibberellins promote tobacco seed germination by distinct pathways
Planta in press (2001)
Comment: These two paper present evidence that plant brassinosteroid (BR) hormones play a role in promoting seed germination. Abscisic acid (ABA) inhibits and gibberellins (GA) promote germination of Arabidopsis thaliana and Nicotiana tabacum, but only little was known about the effect of BR on seeds. In their work with Arabidopsis Camille M. Steber (United States Department of Agriculture-Agricultural Research Service, Washington State University, Pullman, Washington) and Peter McCourt (Department of Botany, University of Toronto, Ontario, Canada) show that BR rescues the germination phenotype of severe GA-biosynthesis mutants and of the GA-insensitive mutant sleepy1. In my work with non-photodormant tobacco seeds I show that while GA can replace light in promoting dark germination, BR promotes tobbacco germination in the light and in the dark. GA, but not BR, can break photodormancy of photodormant tobacco seeds. In both species BR counteracts the inhibitory effect of ABA on seed germination in an GA-independent manner. Germination of the BR-deficient det2-1 and the BR-insensitive bri1-1 mutant is more strongly inhibited by ABA compared to Arabidopsis wild-type. Taken together these results suggest that BR and GA promote seed germination by distinct signalling pathways and that BR might act by direct promotion of the embryo's growth potential.
|
 |
|
| December 2000 |
Beaudoin N, Serizet C, Gosti F, Giraudat J. Interactions between abscisic acid and ethylene signaling cascades.
The Plant Cell 12: 1103-1115 (2000)
Ghassemian M, Nambara E, Cutler S, Kawaide H, Kamiya Y, McCourt P. Regulation of abscisic acid signaling by the ethylene response pathway in Arabidopsis.
The Plant Cell 12: 1107-1126 (2000)
Comment: Seed germination is inhibited by abscisic acid (ABA) and in many species promoted by ethylene (reviewed by Kepczynski and Kepczynska, Physiol. Plant. 101: 720-726, 1997). The work presented in the two Plant Cell papers demonstrate with Arabidopsis mutants that the two signaling pathways interact during germination. Ethylene appears to promote germination by decreasing the sensitivity to endogenous ABA and therefore seems to be a negative reulator of ABA action during seed germination. ABA also inhibits the transcipt expression of ethylene-responsive element binding proteins (EREBPs) in germinating tobacco seeds (Leubner-Metzger et al., 1998). The ethylene-inducible EREBPs seem to be another seed-associated step where the ABA and ethylene signaling pathways interact.
|
 |
|
| November 2000 |
Weber H, Rolletschek H, Heim U, Golombek S, Gubatz S, Wobus U. Antisense-inhibition of ADP-glucose pyrophosphorylase in developing seeds of Vicia narbonensis moderately decreases starch but increases protein content and affects seed maturation.
The Plant Journal 24: 33-43 (2000)
Comment: During seed development, processes like cell elongation, differentiation and storage activities appear to be subject to metabolic control (reviewed by Wobus and Weber, Biol Chem 380: 937-944, 1999). The group of Hans Weber and Ulrich Wobus from the Institute for Plant Genetics and Crop Plant Research (IPK) in Gatersleben (Germany) showed that reduction of the small subunit of the ADP-glucose pyrophosphorylase by antisense transformation not only affected starch content, seed filling and other storage activities, but most interestingly extended the seed maturation phase. They conclude that alteration in carbon metabolism can induce temporal changes in seed development.
|
 |
|
| October 2000 |
Nonogaki H, Gee OH, Bradford KJ. A germination-specific endo-ß-mannanase gene is expressed in the micropylar endosperm cap of tomato seeds
Plant Physiology 123: 1235-1245 (2000)
Toorop PE, van Aelst AC, Hilhorst HWM. The second step of the biphasic endosperm cap weakening that mediates tomato (Lycopersicon esculentum) seed germination is under control of ABA.
Journal of Experimental Botany 51: 1371-1379 (2000)
Comment: These papers represent recent progress concerning the role of hydrolases during endosperm weakening of germinating tomato seeds. The group of Henk Hilhorst from Wageningen University (The Netherlands) proposed that tomato endosperm cap weakening is a biphasic precess. The first phase is characterized by major ABA-independent weakening, whereas the second phase just prior to radicle emergence is under the control of ABA. The group of Kent Bradford from the University of Califormia, Davis (California, USA) cloned a cDNA (LeMAN2) encoding a tomato endo-ß-mannanase. LeMAN2 expression is confined to the endosperm cap tissue of seeds prior to radicle emergence, is induced by GA in gib-1 mutant seeeds, but is not inhibited in wild-type seeds by ABA. These results support the view that the LeMAN2 germinative mannanase is one component among several that are required for cell wall disassembly and tissue weakening in the endosperm cap to allow radicle emergence. The final ABA-dependent step of radicle emergence might involve the expression of additional ABA-controlled hydrolase genes. The role of endo-ß-mannanase during seed germination is reviewed by J. Derek Bewley (Trends in Plant Science 2: 464-469, 1997).
|
 |
|
| July 2000 |
Leubner-Metzger G, Meins F Jr. Sense transformation reveals a novel role for class I ß-1,3-glucanase in tobacco seed germination.
The Plant Journal 23: 215-221 (2000)
Comment: 'Coat-enhanced' seed dormancy of many dicotyledonous species is released during afterripening. In the case of endosperm-limited germination, e.g. in lettuce and members of the Solanaceae family, it is believed that hydrolytic enzymes facilitate weakening of the endosperm surrounding the radicle tip by hydrolysing cell-wall materials; however, direct evidence for this hypothesis from studies with transgenic seeds was lacking. In my research project in the group of Fred Meins at the Friedrich Miescher Institute, Basel (Switzerland) I established by sense transformation that class I ß-1,3-glucanase ( ßGLU I) has a causal role in endosperm rupture. Treating afterripened tobacco seeds with abscisic acid (ABA) delays endosperm rupture and inhibits ßGLU I induction. Sense transformation with a chimeric ABA-inducible ßGLU I transgene resulted in overexpression of ßGLU I in seeds and promoted endosperm rupture of mature seeds and of ABA-treated afterripened seeds. Taken together, these results provide direct evidence that ßGLU I contributes to endosperm rupture. Overexpression of ßGLU I during germination also replaced the effects of afterripening on endosperm rupture. This suggests that regulation of ßGLU I by ABA signaling pathways might have a key role in afterripening.
|
 |
|
| February 2000 |
Debeaujon I, Léon-Kloosterziel KM, Koornneef M. Influence of the testa on seed dormancy, germination, and longevity in Arabidopsis.
Plant Physiol 122: 403-413 (2000)
Debeaujon I, Koornneef M. Gibberellin requirement for Arabidopsis seed germination is determined both by testa characteristics and embryonic abscisic acid.
Plant Physiol 122: 415-424 (2000)
Comment on both publications: The characteristics of the testa (seed coat) are important for the coat-enhanced dormancy of Arabidopsis seeds.The group of Maarten Koornneef at the Laboratory of Genetics, Wageningen University (The Netherlands) analyzed the function of the testa by comparing mutants that often exhibited reduced dormancy. Both structural and pigmentation testa mutants deteriorated faster than the wild types during natural aging at room temperature, with structural mutants being the most strongly affected. The mechanisms imposing a gibberellin (GA) requirement to promote the germination of dormant and non-dormant Arabidopsis seeds were analyzed using the GA-deficient mutant ga1, several testa pigmentation and structure mutants, and the abscisic acid (ABA)-deficient mutant aba1. Interestingly, testa mutants, which exhibit reduced seed dormancy, were not resistant to GA biosynthesis inhibitors and exhibited enhanced GA-sensitivity. These results suggest that GAs are required to overcome the germination constraints imposed both by the testa and ABA-related embryo dormancy.
|
 |
|
January 2000
|
Grappin P, Bouinot D, Sotta B, Miginiac E, Jullien M. Control of seed dormancy in Nicotiana plumbaginifolia: post-imbibition abscisic acid synthesis imposes dormancy maintenance.
Planta 210: 279-285 (2000)
Comment: Abscisic acid (ABA) is needed to establish and maintain primary dormancy in many species including the genus Nicotiana. The onset of dormancy in Nicotiana seeds is correlated with a peak in ABA content, which declines rapidely during further seed maturation (DAP 15-25 in tobacco). The work of the group Marc Jullien at the INRA in Versailles (France) demonstrates that afterripening of Nicotiana seeds is correlated with a further decline in ABA content and decreased sensitivity to ABA. Dry dormant (fresh) seeds contain more ABA than dry non-dormant (afterripened) seeds. Interestingly de novo ABA synthesis was found in imbibed dormant seed, but not in non-dormant seed. Gibberellin (GA) treatment inhibited the accumulation of ABA in dormant seeds. This work reveals an important role for ABA synthesis in dormancy maintenance in imbibed seeds.
|
|
 |
| |
|
|
|

|
 |

