The Plant Cell 21: 3803-3822 (2009)
Ethylene interacts with abscisic acid to regulate endosperm rupture during germination: a comparative approach using Lepidium sativum and Arabidopsis thaliana [W][OA]
Warwick Horticulture Research International (HRI), Warwick University, Wellesbourne, Warwick CV35 9EF, United Kingdom (Ka.M., C.S.C.C., J.R.L., W.E.F.-S.)
Palacky University and Institute of Experimental Botany Academy of Sciences of the Czech Republic, Laboratory of Growth Regulators, CZ-78371 Olomouc, Czech Republic (V.T., M.S.)
Université Pierre et Marie Curie-Paris 6, Germination et Dormance des Semences, UR5, Site d'Ivry, F-75005 Paris, France (F.C.)
Received July 23, 2009; Returned for revision October 12, 2009; Accepted November 17, 2009; Published December 18, 2009
www.plantcell.org/cgi/doi/10.1105/tpc.109.070201
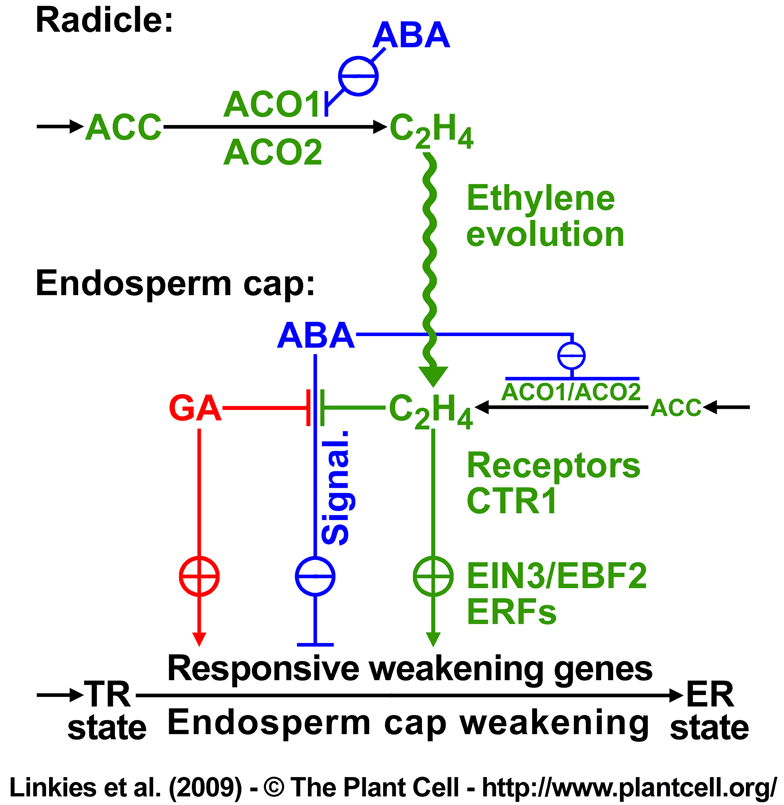
Figure 10. Proposed model for the hormonal regulation of endosperm cap weakening and rupture.
According to our working model, endosperm cap weakening is required during the transition from testa rupture (TR) to endosperm rupture (ER), and endosperm cap weakening in turn requires ethylene biosynthesis and signaling. Endosperm cap weakening is a developmental process that is regulated by interactions between the cap and the radicle. GA, as an embryo signal, releases coat dormancy (if present) and induces the cap weakening process. Thereafter, weakening is a cap-autonomous process, and the rate of this process is regulated by the GA-ABA and ethylene-ABA antagonisms. The involvement of GA as antagonist of ABA in seed germination is well established in different Brassicaceae (e.g., Yamaguchi et al., 2001; Ogawa et al., 2003; Chiwocha et al., 2005; Müller et al., 2006; Iglesias-Fernandez and Matilla, 2009).
GA biosynthesis may be more important in the early phase of germination, and ethylene biosynthesis may be more important for the late phase of the process. Ethylene biosynthesis in germinating seeds is regulated differently in the radicle and the endosperm cap, and this effect is mediated by ACO2 gene expression in the following way: although both ACO2 and ACO1 are active in the radicle and endosperm cap, the total activity is greater in the former. The radicle produces ethylene in excess, and this is targeted at the endosperm cap. ABA delays the ACO activity in the radicle and inhibits ACO1 transcript accumulation, but ABA does not inhibit ACO2 transcript accumulation. The later increase in ACO activity in the radicle of ABA-treated seeds is therefore due to ACO2, and the ethylene produced promotes endosperm cap weakening by antagonizing the ABA inhibition. In the endosperm cap, ABA inhibits ACO2 and ACO1 transcript accumulation. A basal level of ethylene signaling is required to maintain ACO2 transcript levels in seeds.
Ethylene does not affect the seed ABA levels and therefore must counteract the ABA-induced inhibition of endosperm rupture by interfering with ABA signaling. High degrees of GA and ethylene sensitivity of the endosperm cap are associated with the after-ripened (post coat dormancy) seed state and are prerequisites for ethylene-enhanced expression of ABA-inhibitable downstream weakening genes in the cap. The products of the weakening genes cause endosperm cap weakening by cell wall loosening and cell separation that finally leads to endosperm rupture and radicle emergence.
Synopsis: Tissue weakening of the endosperm is required to allow radicle protrusion during seed germination. Cross-species work including tissue-specific transcriptome analysis and biomechanical measurement of endosperm weakening provided a new mechanistic model that explains how ethylene promotes seed germination and counteracts the inhibition of endosperm cap weakening by abscisic acid.
| Article in PDF format (1.8 MB) Supplemental data file (1.8 MB) Abstract of Plant Cell 2009 |
|
|
|
The Seed Biology Place |
Webdesign Gerhard Leubner 2000 |